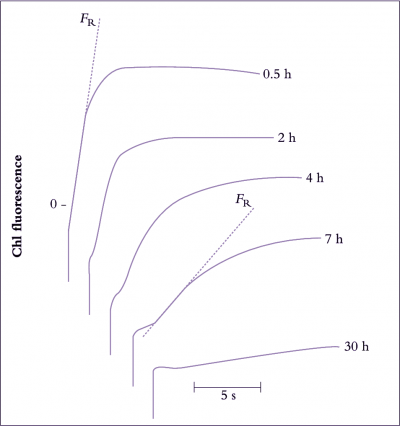
Figure 14.18. Changes with time in in vivo chlorophyll a fluorescence induction kinetics in response to chilling. In chilling studies, chlorophyll fluorescence has commonly been measured as the rate of rise in fluorescence yield induced by illuminating dark-adapted tissue (the FR value). In isolated chloroplasts, a decrease in photosystem II activity is correlated with a decrease in FR (Smillie and Nott 1979). This figure shows the progressive decline in FR in a trifoliate bean (Phaseolus vulgaris L. cv. Canadian Wonder) leaflet chilled at 0°C in darkness. The first measurement of FR was made after allowing 30 min at 0°C for temperature equilibration of the leaflet. The measurement was repeated on the same area of the leaflet at the times indicated on the figure. The longer the time of` chilling, the greater the degree of chilling injury, and the slower the rate of rise of FR. The greater the chilling sensitivity of a cultivar, the shorter the time taken for a 50% decrease in FR (Original data courtesy R.M. Smillie)