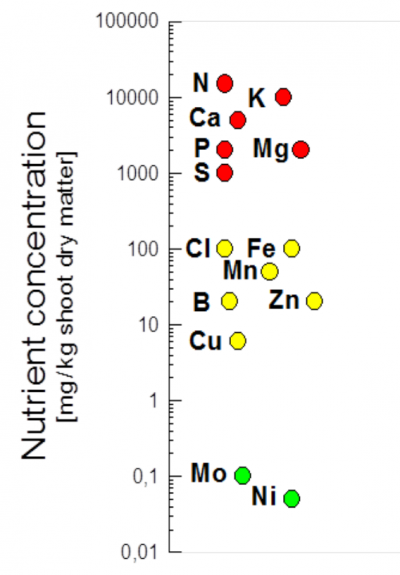
Figure 4.2 Mineral nutrient concentrations in plant leaves. Concentrations can be larger or smaller than shown here. Note, lower concentrations occur in roots and wood. (Adapted from Marschner 1995)
Although the absolute quantities of nutrients required vary between plant species, genotypes and growth environments, essential nutrients are categorised into so-called macronutrients (N, K, Ca, P, Mg, S) that plants require in larger quantities, and micronutrients (Fe, Cl, Mn, B, Zn, Cu, Mo, Ni) that are needed in small amounts (Figure 4.2). Additional beneficial elements include Si (e.g. for grasses) and Na (for many sea-shore plants).
Macronutrients form the structural components of proteins, cell walls, membranes, nucleotides and chlorophyll, and have roles in energy and water maintenance. The macronutrient potassium has a special function in regulating the osmotic potential of plant cells. Under saline or dry conditions, Cl (and for some plants Na) is important in plant water relations.Micronutrients mainly provide functional groups in enzymes (BOX 1 shows how Ni forms the active site in urease, as an example).
BOX 1 – Nickel (Ni) at the centre of UreaseIn 1926 James B. Sumner from Cornell University studied the structure of Urease from Jack Bean plants and demonstrated that the enzyme is also a protein. This work led to the recognition that most enzymes are in fact proteins and in 1946 Sumner was awarded the Nobel Prize in Chemistry. Urease is an enzyme that breaks down urea to ammonia and carbon dioxide in plants, bacteria and fungi and contains a nickel active site. (NH2)2CO + H2O -> CO2 + 2NH3 For 3D structure see http://www.proteopedia.org/wiki/index.php/Urease Further reading: Follmer 2008; Carter et al. 2009. |
In terrestrial ecosystems and in agriculture, the availability of nitrogen (N) and phosphorus (P) are often limiting and so affect plant growth and productivity most strongly. However, other elements can also be limiting. Plants showing nutrient deficiencies will exhibit symptoms such as stunted growth, leaf or shoot tip chlorosis, and defoliation, and will die if supplements are not provided. Fertilisers are applied to supply essential elements in agriculture to maximise plant growth and enhance yields. Along with the discovery of ‘dwarfing genes’ and development of short stature crop varieties, it was the use of synthetic nitrogen fertilisers that played a significant role in the Green Revolution of the 1930s-1960s.
The acquisition of mineral nutrients starts with their movement from the surrounding soil to root surfaces. The movement of nutrients from bulk soil towards plant roots occurs via diffusion or mass flow (Figure 4.3). Root interception occurs as the root comes in contact with, and displaces the soil through which it is growing. Nutrient availability in soils and the physical and chemical factors influencing their movement from soils to the root surface is comprehensively described in a review by Marschner and Rengel (2012). Nutrients are taken up into roots by active or passive transport across cell membranes, which is described later in this chapter (Section 4.5).
Diffusion occurs along a concentration gradient, over relatively short distances (in the order of 1 cm). As roots take up nutrients and ions from the soil a depletion zone can be established allowing diffusion to occur into the depletion zone. The rate of diffusion depends on how fast the roots are taking up the nutrient, how much of the nutrient is present in the soil (this determines the steepness of the concentration gradient that forms) and also on the mobility of the ions by diffusion. Soluble ions would take about a day to diffuse 1 cm; ions bound to the soil matrix would take longer. For examples, Marscher and Rengel (2012) show that nitrate by diffusion in a ‘typical’ soil travels 3 mm in a day, potassium about 1 mm in a day, and phosphate moves only about 0.1 mm in a day. This illustrates the importance of root hairs in intercepting and accessing phosphate.
Mass flow is driven by the uptake of water caused by the transpiration rate of the plants and can occur over long distances. Many soluble nutrients such as nitrate are dissolved in the soil water and as the plant pulls the water from the soil, the nutrients move too. Some nutrients move by mass flow faster than their uptake rate so they build up on the surface of the root during daylight hours (Marschner and Rengel, 2012). The rate of movement by mass flow of solution depends basically on the rate of transpiration of the plant, so there is little movement at night. It is also influenced by soil water content and soil texture (see Chapter 3, Section 3.4).
Nutrients are unevenly distributed in the soil, generally being concentrated in the topsoil due to decomposition of leaf litter, but also dispersed elsewhere in pockets. Uneven surface enrichment arises from diverse sources such as dead fauna, urine patches from grazing animals, and localised application of fertiliser. Phosphorus and all cations are relatively immobile as they bind to the soil while nitrate and other anions (except phosphorus) are soluble and can readily be leached to deeper soil layers.
Because the soil is so heterogeneous, plants have developed adaptable (plastic) root systems so that the roots proliferate close to the nutrients for uptake.