(a) General characteristics
Any hypothesis of phloem loading must account for the following characteristics:
- Elevated solute concentration in se–cc complexes. Estimated solute concentrations in sap of se–cc complexes is much higher than concentrations in sap of surrounding cell types, irrespective of whether phloem loading is by an apoplasmic or a symplasmic route.
- Selective loading of solutes into se–cc complexes. Chemical analysis of phloem sap by techniques shown above in Section 5.2 reveals relative solute concentrations different from those in surrounding cells. Phloem loading is therefore a selective process.
(b) Symplasmic loading
The above-described characteristics have been used to argue against loading of se–cc complexes through a symplasmic route on the grounds that plasmodesmata lack mechanisms for concentrating and selecting solutes. However, a contribution of plasmodesmata to concentrating and selecting solutes cannot be precluded from our current knowledge of plasmodesmal structure and function.
Plants that load se–cc complexes through a symplasmic route translocate 20–80% of sugars in the form of raffinose-related compounds such as raffinose, stachyose and verbascose (Section 5.2.3(c)). Grusak et al. (1996) proposed a model for symplasmic phloem loading that accounts for the general characteristics stated above. According to this model (Figure 5.16), sucrose diffuses from mesophyll and bundle sheath cells into intermediary (companion) cells through plasmodesmata. Within companion cells, sucrose is thought to be enzymatically converted to oligosaccharides (raffinose or stachyose) maintaining a diffusion gradient for sucrose from mesophyll cells into se–cc complexes. The molecular-size-exclusion limit of plasmodesmata interconnecting mesophyll and companion cells is such that it prevents back diffusion of stachyose and raffinose molecules, which are larger than sucrose. These oligosaccharides are able to diffuse through plasmodesmata with larger diameters linking companion cells with sieve elements (van Bel 1993). This model accounts for selective loading of sugars to achieve high photoassimilate concentrations in phloem elements.
Fig_p_5.25.png
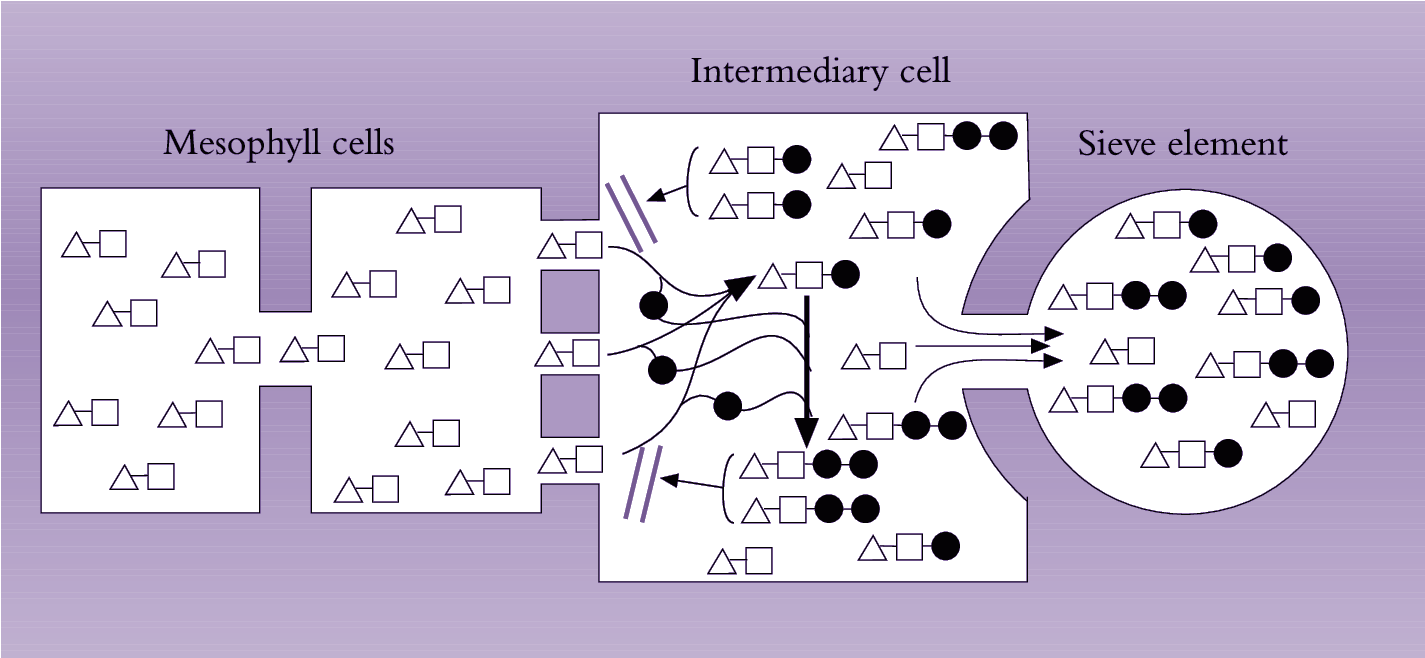
Figure 5.16. Model of the ‘polymerisation trap mechanism’ to explain symplasinic phloem loading against a solute concentration gradient. Sucrose moves through a symplasmic path from photosynthetic cells into intermediary (companion) cells of the minor veins. Sucrose movement is by diffusion down a concentration gradient maintained by the polymerisation of sucrose into oligosaccharides (raffinose and stachyose) in intermediary cells. Diffusion of these oligosaccharides into mesophyll cells is prevented, as their size exceeds the molecular exclusion limit of plasmodesmata joining mesophyll and intermediary cells. However, the larger-diametered plasmodesmata linking intermediary cells with sieve elements permit oligosaccharides to be loaded into sieve elements for export from the leaf. [], glucose; Δ, fructose; • galactinol (Based on Grusak et al. 1996)
(c) Apoplasmic loading
Phloem loading with an apoplasmic step is an attractive model, explaining both how solutes become concentrated in se–cc complexes (energy-coupled membrane transport) and how they could be selected by specific membrane transporters (see van Bel 1993). Identifying transport mechanisms responsible for photoassimilate transport to and from the leaf apoplasm has proved challenging.
Based on estimates of sucrose fluxes and high sucrose concentrations in phloem sap, there is little doubt that sucrose loading into phloem is energy dependent. The demonstration that PCMBS blocks loading of photoassimilates in whole leaves of certain species (Section 5.3.2(b)) points to carrier-mediated transport across plasma membranes. Genes encoding sucrose porters have been cloned from leaf tissue (Frommer et al. 1996) and shown to be specifically expressed in leaf phloem. Complementation studies in yeast defective in sucrose transport suggest that the phloem-located sucrose porter catalyses sucrose/proton symport in a similar way to that illustrated in Figure 5.32. Antisense transformants of potato with low abundance of this symporter have impaired sucrose transport (Section 5.32(b)).
In contrast to photoassimilate uptake from phloem apoplasm, very little is known about the mechanism of sugar efflux into the apoplasm until very recently. Estimates of photoassimilate flux to phloem apoplasm, based on rates of sucrose export from leaves, suggest that this transport event must be facilitated by other transport processes (van Bel 1993). This is now confirmed by the recent cloning of sucrose efflux protein that sheds a light on the molecular mechanisms of phloem loading (Chen et al. 2012).
