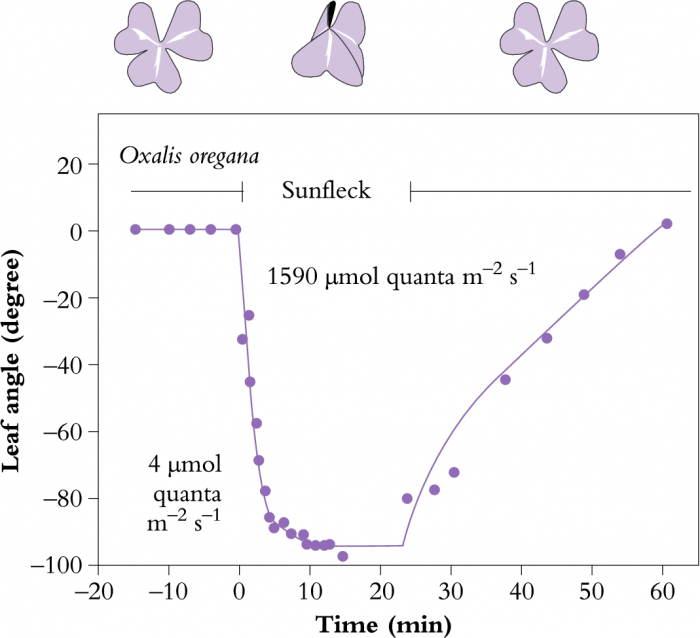
Many plants can change their leaf angles and orientation in response to a change in light. For some, to increase interception while for others, to avoid high light. A good example of optimising light interception through leaf movement is given by Oxalis oregana, an understorey herb of redwood forests in western USA (Figure 12.4). This plant is able to track sunlight on dull days, but can change leaf angle from horizontal to vertical in only 6 min, if exposed to full sunlight. In this way, leaves can maintain maximum photosynthetic rates under a variety of light conditions but can avoid photoinhibition of photosynthesis by leaf folding. Omalanthus novo-guinensis, an Australian rainforest plant, can also change leaf angles within about 20 min in response to full sunlight (Watling et al. 1997b).
Another way of reducing light capture is a change in leaf-surface properties. Many plants in high light environments have a high reflectance of their leaves from a coat of hairs or wax or even salt crystals. Cotyledon orbiculata, a crassulacean acid metabolism (CAM) plant from southern Africa, produces a wax coating on the leaves. Plants grown at high light produce copious quantities of white wax which reflects 60% of incident light whereas plants grown in low light produce very little wax and leaf reflectance drops to 9% (Figure 12.5). Young eucalypt leaves also produce wax, while leaves of Celmisia longifolia, the snow daisy of the Australian Alps, are covered in a thick layer of silvery fibres. In these instances, plants are avoiding high light by creating their own shade, but does leaf anatomy adjust to environments where light is limiting?
Epidermal cells in some rainforest shade-adapted species are shaped to enhance light capture by acting as a lens. The optical properties of such cells focus incident sunlight into the layer of photosynthetic tissue just below the epidermis, reducing light lost due to reflectance and transmittance.
Light interception can also be regulated at a tissue and organelle level. Photosynthetic tissue can be concentrated equally on both sides of a leaf (isobilateral) to maximise light absorption from either side, or preferentially on one side (dorsiventral) as is common in species where leaves are predominantly horizontal.
Chloroplast density and location within leaves is also sensitive to the light climate, and energy capture varies accordingly. Alignment along vertical cell walls will reduce overall absorption of incident light, and in Oxalis leaves absorbance can be reduced 20% when chloroplasts align less to the horizontal and more to the vertical walls of mesophyll cells.
Once sunlight has been intercepted by an assimilatory organ, photon absorption then depends on the extent and nature of light-absorbing pigments in the photosynthetic tissues. In terrestrial plants, the major light-absorbing pigments are chlorophylls a and b plus a range of carotenoids which can act as accessory pigments. Compared with high-light plants, plants grown in low light tend to allocate relatively more resources to their light-harvesting pigments and the associated proteins than to the enzyme Rubisco and other soluble proteins involved in CO2 fixation. This shift in allocation of nitrogen-based resources can be accompanied by marked changes in leaf anatomy, especially depth of mesophyll tissue (see Case study 12.1) and reflects a need for increased efficiency of light absorption when sunlight is limited.
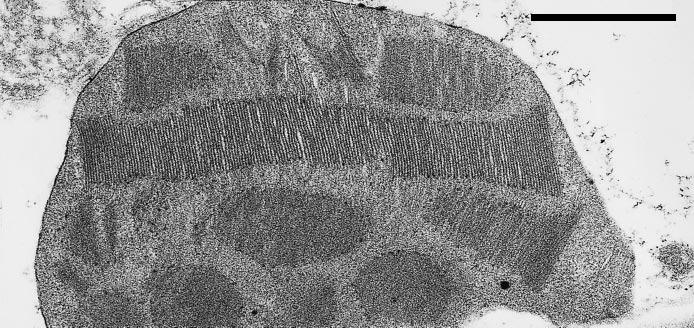
There are also differences in chloroplast structure between plants grown in low light and high light. Shade chloroplasts tend to be larger than those found in sun plants. They also contain more thylakoid membranes which show higher levels of randomly arranged granal stacking into appressed regions, as shown by the extreme development of grana in Figure 12.6. The higher proportion of appressed to non-appressed membranes found in shade chloroplasts is the result of increased photosynthetic system II (PSII) and antenna (LHCII) content. LHCII is thought to be involved in thylakoid appression and formation of granal stacks. Plants grown in low light also tend to have lower Chl a/b ratios. Chlorophylls a and b are both associated with the light-harvesting antennae, while only Chl a is found in the reaction centres. A lower a/b ratio, therefore, reflects an increase in LHCII complexes relative to reaction centres (see Chapter 1, Section 1.2).
In addition to differences in leaf anatomy and chloroplast fine structure, energy derived from absorbed sunlight is processed in ways that differ subtly between shade-grown and sun-grown plants. In high light, there is a requirement for greater capacity in both the light and CO2 fixation reactions of photosynthesis. Photosynthesis–light response curves for shade and sun plants (Figure 12.7) illustrate such differences. The initial slope of each light response curve represents the quantum or photon efficiency of photosynthesis. This is the same for sun and shade plants. The reason it does not change is that the efficiency of the light reactions is the same irrespective of how much light has been received during growth (i.e. eight photons are required for the evolution of one molecule of O2 and fixation of one molecule of CO2 in all plants).
12.1-Ch-Fig-12.7.png
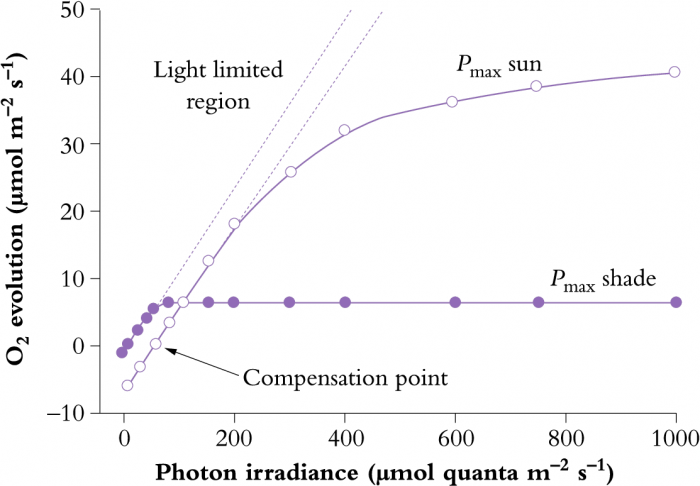
Figure 12.7 Photosynthesis-light response curve for typical shade and sun plants, showing relationships between photosynthetic rate and absorbed light (expressed as a photon irradiance). Dashed lines are extrapolations of initial linear slopes where photosynthesis is light limited, and represent quantum yield (moles of O2 evolved per mole quanta absorbed). (Original data courtesy S.A. Robinson)
However, sun plants tend to have a greater capacity for photosynthetic electron transport (greater abundance of transport components such as Cyt b559, Cyt b563, Cyt f and plastoquinone). They also have a greater capacity for ATP synthesis per unit of chlorophyll compared with shade plants. Taken together, these capacities of sun plants allow more sunlight to be processed into ATP and NADPH for use in CO2 assimilation and other synthetic events. Such capacity is also matched by a greater investment in enzymes of the photosynthetic carbon reduction (PCR) cycle, resulting in a higher light-saturation point and a higher maximum rate of photosynthesis (Pmax) for sun plants (Figure 12.7). As a further distinction, sun leaves tend to be thicker and have more cell layers. They also have higher stomatal conductances to facilitate rapid uptake of CO2.
A higher photosynthetic capacity in sun plants does, however, incur some costs. The sun leaves tend to have higher respiration rates which increases the light-compensation point relative to shade plants (Figure 12.7). Higher respiration rates probably result from (1) increased carbohydrate processing in high light, (2) increased costs of constructing sun leaves and (3) a higher cost of maintaining sun leaves. Further details on maintenance costs are given in Chapter 5, Section 6.5.
Greater transpiration is a further cost of the higher photosynthetic capacity as a result of higher stomatal conductance. Sun plants often respond to the greater transpiration by increasing their root : shoot ratios. Under conditions where water is limiting, however, stomatal conductance may be reduced, sacrificing photosynthesis in favour of slower transpiration.