M. Watt
4.1-CS-Fig-1.png
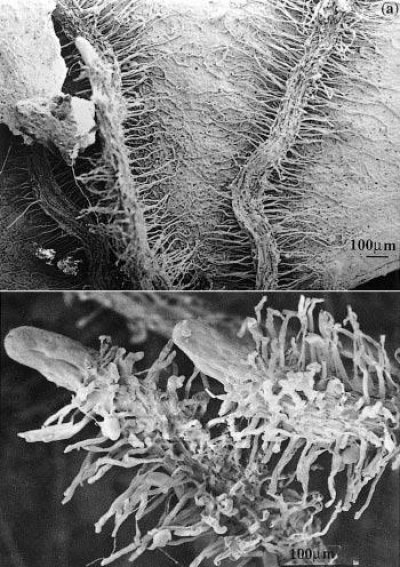
Figure 1 Cluster roots in Banksia serrata growing on Hawkesbury Sandstone hillslopes in the Sydney region. a, Roots that have grown across a dead eucalypt leaf extract nutrients remaining in the decaying leaf. b, Clusters of fine rootlets at the tips of roots increase the surface area for nutrient extraction from surrounding soil. Scale bar = 100 µm. (Scanning electron micrograph courtesy S. Gould)
Australian soils generally contain low concentrations of plant-available phosphate, much of it bound with iron–aluminium silicates into insoluble forms or concentrated in the remains of decaying plant matter. Because very little of this phosphate is soluble, most roots extract it only slowly. Plants with cluster roots gain access to fixed and organic phosphate through an increase in surface area and release of phosphate-solubilising exudates. Hence plants with cluster roots grow faster on phosphate-fixing soils than species without clusters.
Cluster roots have a distinct morphology. Intense proliferation of closely spaced, lateral ‘rootlets’ occurs along part of a root axis to form the visually striking structures. Root hairs develop along each rootlet and result in a further increase in surface area compared to regions where cluster roots have not developed.
In the Proteaceae, clusters generally form on basal laterals so that they are abundant near the soil surface where most nutrients are found. For example, Banksia serrata produces a persistent, dense root mat capable of intercepting nutrients from leaf litter and binding the protecting underlying soil from erosion (Figure 1a). New clusters differentiate on the surface of this mat after fires and are well placed to capture nutrients. In contrast, Banksia prionotes forms ephemeral clusters which export large amounts of nutrients during winter. Lupinus albus has more random clusters which appear on up to 50% of roots (Figure 2).
4.1-CS-Fig-2.png
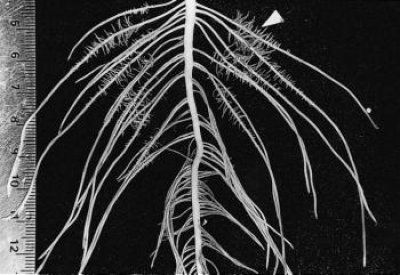
Figure 2 Basal roots of a two-week-old Lupinus albus plant grown in nutrient culture with 1 µM phosphate. Proteoid roots have emerged along the primary lateral roots (arrowhead) and the oldest proteoid rootlets have reached a determinate length of 5 mm. As rootlets approach their final length, they exude citrate for 2-3 d. (mm scale on left side) (Photograph courtesy M. Watt)
Rootlets not only represent an increase in surface area but also exude protons and organic acids, solubilising phosphate and making it available for uptake (Watt and Evans 1999a). Exudates from cluster roots represent up to 10–23% of the total weight of an L. albus plant, suggesting that they constitute a major sink for photoassimilates. However, not all this additional carbon comes from photosynthesis because approximately 30% of the carbon demand of clusters is met by dark CO2 fixation via phosphoenolpyruvate carboxylase. Because cluster roots form on roots of L. albus even when phosphate supply is adequate, growth of L. albus in soils with low phosphate availability is not restricted by an additional carbon ‘drain’ to roots. On the other hand, the great many species which produce cluster roots in response to environmental cues like phosphate deficiency might experience a carbon penalty to support these roots.
Cluster roots on L. albus are efficient with respect to carbon consumption by generating citrate on cue. Most of the citrate exuded by clusters is released during a two to three day period when the cluster is young (Watt et al. 1999b). A large root surface area in clusters works in concert with this burst of exudation to solubilise phosphate before it is re-fixed to clay surfaces or diffuses away.
Cluster roots can mine a pocket of phosphate-rich soil which would otherwise not yield its nutrients. They are an elegant adaptation of root structure and biochemistry to nutrient-poor soils.
References
Dinkelaker B, Hengeler C, Marschner H (1995) Distribution and function of proteoid roots and other root clusters. Bot Acta 108: 183–200
Lamont BB (1993) Why are hairy root clusters so abundant in the most nutrient impoverished soils of Australia? Plant Soil 156: 269–272
Purnell HM (1960) Studies of the family Proteaceae 1. Anatomy and morphology of the roots of some Victorian species. Aust J Bot 8: 38–50
Watt M, Evans JR (1999a) Proteoid Roots. Physiology and Development. Plant Physiol 121: 317–323
Watt M, Evans JR (1999b) Linking development and determinacy with organic acid efflux from proteoid roots of Lupinus albus L. grown with low phosphorus and ambient or elevated atmospheric CO2 concentration. Plant Physiol 120: 705–716
